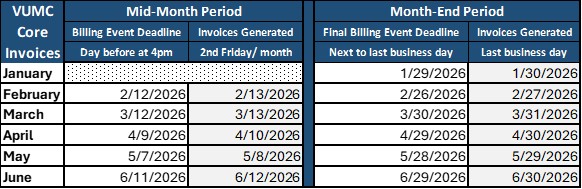
Learn more about VUMC core services by clicking each core name in the list below. Information about VUMC's innovative support model for core facilities can be found in the Core Administration & Policies section of this website. For any questions, please contact Susan Meyn in the Office of Research.
Learn about Vanderbilt University core administration
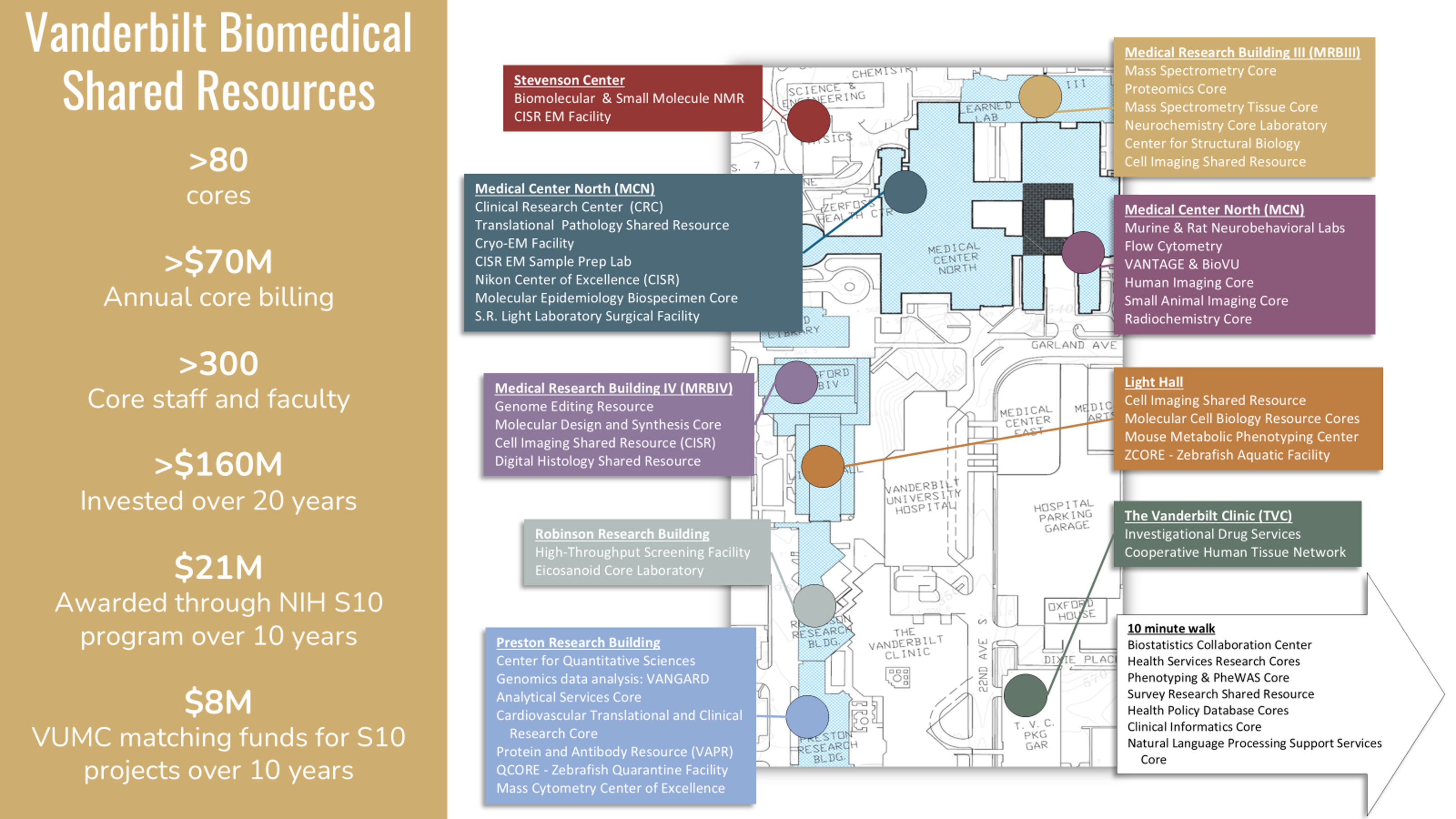
Vanderbilt University School of Medicine - VUMC
Institutional Shared Resources and Core Facilities
A comprehensive list of VUMC cores, including departmental and limited service cores, can be found HERE.
For a list of VU Basic Sciences Shared Resources and Core Facilities, scroll down to end of the list.
-
The Biostatistics Collaboration Center (BCC) provides, enhances, and/or facilitates statistical collaborations involving the design, conduct, analysis or publication of biomedical research at the university. The BCC is comprised of biostatisticians and computer systems analysts from the Department of Biostatistics who are available to work with faculty on a variety of projects. They offer a wide range of highly trained experts with unique expertise for almost any collaboration. The BCC has considerable expertise in the design, conduct, and analysis of large scale clinical trials and research design for basic biomedical research. Varying levels of expertise are also available for consultation, from bachelor’s and master’s level trained biostatisticians and computer systems analysts to full professors.
Center Manager: Sandra Hewston
Email: sandra.hewston@vumc.org
vumc.org/biostatistics/biostatistics-collaboration-center-bcc
vumc.org/biostatistics/vanderbilt-department-biostatistics
-
BioVU provides enabling resource for exploration of the relationships among genetic variation, disease susceptibility, and variable drug responses, and represents a key first step in moving the emerging sciences of genomics and pharmacogenomics from research tools to clinical practice. A major goal of the resource is to generate datasets that incorporate de-identified information derived from medical records and genotype information to identify factors that affect disease susceptibility, disease progression, and/or drug response.
BioVU partners closely with VANTAGE and VANGARD (see below) to generate and analyze genomic data from BioVU DNA samples.
Manager: Laura Jacobus, MHA CCRC
E-Mail: biovu@vumc.org
Website: starbrite.app.vumc.org/vdr/biovu
-
The purpose of the Vanderbilt Clinical Research Center is to provide the resources needed by clinical and translational investigators to carry out their research in an environment optimized for safety, comfort and convenience of the patient or volunteer, with the facilities and expertise to provide the most advantageous milieu in which to address the investigator's experimental requirements. This includes outpatient space, inpatient beds, laboratories, equipment and supplies for clinical research by the faculty of Vanderbilt University, Meharry Medical College, and their collaborating investigators. The use of the center is justified on the basis of the quality and significance of the research and the special need for the Center's facilities.
In the process of achieving its primary objective, the Clinical Research Center can also serve as a resource for teaching medical, graduate, and other multidisciplinary students, a site for research on the methodology of patient care systems, and an unique environment for the apprenticeship of young clinical investigators. The center is a component of the Vanderbilt Institute of Clinical and Translational Research (VICTR) organized under a Clinical and Translational Award (CTSA) made in 2007 by the National Center for Research Resources and the National Institute of Health.Program Manager: Jennifer Adams
E-Mail: jennifer.s.adams@vumc.org
Websites:
vumc.org/crc/vanderbilt-clinical-research-center
starbrite.app.vumc.org/maincrc
-
The Digital Histology Shared Resource (DHSR) provides large-scale digital archiving and quantitative analysis of histological, immunocytochemical and immunofluorescence staining for tissue sections and tissue microarrays. The Ariol SL-50 automated scanning microscope and the Leica SCN400 Slide Scanner deliver solutions for high-resolution imaging in both bright field and fluorescence. Both instruments have high-capacity robotic autoloading (50 slides for the Ariol SL-50 and 384 slides for the Leica SCN400) making them ideal for large slide cohorts and tissue microarrays. The Leica SCN400 scanner can image normal size as well as large format slides and whole mounts. The associated software packages provide complex algorithms for unbiased, automated image analysis and quantitation of immunostaining in both bright field and fluorescence. In addition, these systems can be utilized as a permanent high-resolution solution for those who need archiving of histological material. The DHSR hosts a convenient, web-based digital slide-viewing environment (Digital Image Hub) for the rapid retrieval, review and annotation of scanned material. This easy to use digital slide box allows researchers to share images among colleagues, both at Vanderbilt and at other institutions, from any web browser while remaining in a digitally secure environment. Built-in conferencing features allow investigators to coordinate multi-site discussions of histological samples.
The DHSR also supports shared usage of a Applied Precision/GE Deltavision Deconvolution Fluorescence microscope equipped with a Weatherstation for extended live cell imaging. The DHSR also houses an Optronix Digital Colony Counter for automated counting and size quantitation of cell colonies grown in multiple culture formats.
Scientific Director: James R. Goldenring, M.D., Ph.D.
Managing Director: Joseph Roland, Ph.D.
E-Mail: joseph.t.roland@vumc.org
Website: vumc.org/dhsr/
-
The Division of Animal Care (DAC) is the service core of the Office of Research that provides procurement, husbandry, and healthcare for experimental animals, as well as scientific/technical support for VU researchers.
Our mission is to provide humane animal care that meets the highest national standards supporting knowledge and discovery through research. To that end, the DAC endeavors to provide excellent animal care and customer service in support of Vanderbilt’s research and teaching programs.
Office hours are 7:30 a.m. - 4:30 p.m. Monday-Friday.
For weekend, holiday and after-hours emergencies, contact the DAC by calling the numbers posted near the phones located in each facility or dialing 615.322.2231 and listening to the message to identify the appropriate individual to call.
E-Mail: DACbilling@vumc.org
Website: vumc.org/acup/ (VUMC or VU password required)
-
The Molecular Epidemiology Biospecimen Core Laboratory provides laboratory services to support molecular and genetic epidemiological studies. Services include biological sample collection and storage, genotyping, and biomarker assays. Study consultations are available to discuss the needs of your project.
Director: Qiuyin Cai, M.D., Ph.D.
Core Manager: Regina Courtney
Website: vumc.org/vec/our-resources/labThe Survey Research Shared Resource (SRSR) offers a wide range of research services available to support investigators at Vanderbilt University needing to conduct survey research. The SRSR provides services to Vanderbilt investigators related to the development and implementation of telephone, web and mixed mode surveys. The SRSR operates a telephone Survey Room with 15 workstations, which are open 7 days a week with shifts spanning both days and evenings. The SRSR is committed to the precise and excellent collection of survey data according to specialized research protocols.
Director: Martha Shrubsole, Ph.D.
Core Manager: John White
Website: vumc.org/vec/our-resources/srsr
-
The Flow Cytometry Shared Resource (FCSR) provides flow cytometry services to faculty, staff, and students across Vanderbilt, VUMC, and external investigators on a fee-for-service basis. The staff maintains all instruments and provides the following services: data acquisition, both by facility staff and qualified users, sorting, instruction in performing data analysis, instruction on instrumentation (analytical and cell sorting), consultation on experimental design, sample handling, staining protocols, education on basic principles of flow cytometry through seminars, lab meetings and course work.
Co-Scientific Directors: Eric Skaar, Ph.D., M.P.H. & Scott Hiebert, Ph.D.
Managing Director: David Flaherty
E-Mail: david.k.flaherty@vumc.org
Website: vumc.org/flowcytometry/
-
HEALTH SERVICES RESEARCH CORES
Qualitative Research Core
Offers consultative services, equipment, and facilitation to support investigators planning to conduct qualitative studies, with a particular focus on use of structured and semi-structured interviews, web-based collection of qualitative data, direct observation of behavior, and focus groups.
Contact: kemberlee.bonnet@vanderbilt.eduImplementation Science/Quality Improvement | Collaborative Shared Resource
Provides consultation and research support services for the implementation and evaluation of initiatives to improve the quality, safety, value, and delivery of health care. Services include consultation on the use of implementation science frameworks, study design (including hybrid designs, quasi-experimental and pragmatic clinical trials), selection of process and outcome measures, program evaluation, and assistance with manuscript and grant preparation.
Contact: robyn.tamboli@vumc.orgDirector: Amanda Mixon, MD, MS
Core Manager: Robyn Tamboli, PhD
Website: vumc.org/implementation/VIQIcoreEffective Health Communication | Collaborative Shared Resource
Provides consultation on aspects of research study design, informed consent, patient recruitment, measurement, data collection, quantitative and qualitative analysis, and manuscript preparation related to health literacy, numeracy, cross-cultural communication and mobile device or internet-based communication.
Contact: erin.m.bergner@vumc.org
-
The Vanderbilt Analytical Services Core facilitates diabetes, endocrine, and metabolic research across a range of species. Sensitive, reproducible, and inexpensive analysis of hormones, lipids, amino acids, stable isotopes, and markers of oxidative stress and inflammation is provided. Investigators are given access to assays which are not readily available, assistance with method development, training, technical support, and access to research space and personnel.
Manager: Dale Edgerton
E-Mail: dale.edgerton@vanderbilt.edu
Website: vumc.org/hormone/
-
The Investigational Drug Service (IDS) is a team of specially trained pharmacists and certified pharmacy technicians within the VUMC Department of Pharmaceutical Services. The IDS team is dedicated to helping investigators accomplish research goals while maintaining compliance with all applicable standards and regulations. Study protocol dispensing can be accommodated for inpatient and outpatient studies on the main campus and at clinic sites such as One Hundred Oaks.
Manager: Donna Torr
E-Mail: donna.k.torr@vumc.org
Website: https://www.vumc.org/department-pharmacy/investigational-drug-services
-
This Molecular Cell Biology Resource Core (MCBR) provides custom DNA and RNA synthesis as well as custom cell culture media at a fraction of the cost of commercial sources. The supply arm of this resource stocks freshly prepared bacteria growth plates with or without antibiotics, transformation competent bacteria, and commercial kits and reagents for molecular biology research. On-line ordering is available and there is free delivery to the lab.
The Bioanalytics Facility houses state-of–the-art instrumentation for imaging and quantitation of fluorescent, radioactive, luminescent, and chemiluminescent signals from gels, membranes, or multiple well plates. Gels and membranes stained with visible dyes or ethidium bromide can also be analyzed. Digitized images can be saved to transportable storage for further analysis in the lab or prints can be made on site. This facility also has four "real-time" PCR instruments available on a sign-out basis.
Manager: Kris Ellis
E-Mail: kris.ellis@vumc.org
-
The core mission is to assist researchers who seek to advance the basic informatics methods, and to apply advanced methods to "understand" unstructured, and sometimes inaccurate, biomedical text and electronic medical record data.
For basic science applications, we provide services for natural language processing methods, terminology development, and integration of disparate data sources (such as coded and free text sources).
We provide three major domains of application of advanced informatics techniques: educational informatics and clinical phenotyping (deriving phenotypes from EMR records). We pursue the latter to discover clinical knowledge, gene-disease relationships (genomics, GWAS, and PheWAS), and gene-drug-outcome relationships (pharmacogenomics).
Core Director (Phenotyping Collaborative Shared Resource): Wei-Qi Wei, MD, PhD
Core Director (PheWAS Core): Lisa BastaracheTo make a service request, please click HERE.
For general questions or specific questions on a service request, please contact phewas_core@vumc.org.
Website: https://www.vumc.org/cpm/phenotyping-and-phewas-core-services
-
The Translational Pathology Shared Resource (TPSR) advances biomedical research and personalized medicine by providing to all Vanderbilt Investigators:
- Access to consented and de-identified human tissue specimens
- Comprehensive, state of the art diagnostic, necropsy and comparative pathology services for animal model systems with specific expertise and focus on mouse models
- Research histology including advanced techniques, immunohistochemistry, laser capture micro-dissection and tissue micro-array preparation
- Access to experts in veterinary and human pathology
Director of Comparative Pathology & Research Histology: Katherine Gibson-Corley, DVM, PhD, DACVP
TPSR Core Manager: Miranda Wilkes
Email: TPSR-Research_Histology@vumc.org
Website: https://www.vumc.org/TPSR/
-
VANTAGE is a genomics core laboratory consolidation initiated by an $8.6 million ARRA funded NIH grant award to Vanderbilt University Medical Center, with an overall goal of creating the VANATAGE collaborative shared resource that accelerates discovery in genome sciences and personalized medicine, providing state-of-the art research shared resource facilities and technology. The NIH grant provided funds for complete modernization of 12,505 Net Square Feet of aging laboratory space in Medical Center North, and for new cutting-edge scientific equipment now in operation.
VANTAGE is a one-stop genomics research resource, including: Illumina genotyping, DNA extraction and banking, Next Generation Sequencing (NGS) and single cell analysis technologies, among many other specialized services. These activities are now part of an overall structure that takes full advantage of the physical co-location together with the Flow Cytometry Shared Resource in VANTAGE. VANTAGE is also the physical laboratory for the BioVU DNA sample bank.Scientific Director: Simon Mallal, MBBS
Assistant Director: Thomas Stricker, MD, PhD
Core Manager (Biobanking/ Genotyping):
Karen Beeri | karen.beeri@vumc.org
Core Manager (NGS Services):
Angela Jones | angela.jones.1@vumc.org
Website: https://www.vumc.org/vantage/home
-
VANGARD works with investigators to design experiments, and analyze and interpret data generated by VANTAGE. The mission of the core is to consolidate the genomics data pipeline across Vanderbilt, allowing investigators to leverage the opportunities provided by next-generation sequencing and other genomics technologies. VANGARD operates in conjunction with VANTAGE, providing experimental design, quality assessment of data, analysis and results interpretation, and data storage to investigators, while VANTAGE provides technical services with a focus on next-generation sequencing including DNA-seq and RNA-seq. VANGARD also provides biostatistical and bioinformatics support for all genomic experiments that utilize BioVU specimens. For small-scale projects, VANGARD uses a fee-for-service model which includes basic experimental design and quantitative analysis for genomic data generated by VANTAGE as well as data storage and backup. Large-scale projects and those that require more complex and detailed analysis may be handled collaboratively.
For questions or to request NGS quotes, please contact Core Manager marisol.a.ramirez@vumc.org
BioVU/VANGARD Liaison: Hui Wu hui.wu@vumc.orgDirector: Yu Shyr, PhD
Technical Director for Informatics: Lynne Berry, PhD
-
The Vanderbilt University Institute of Imaging Science (VUIIS) supports advances in physics, engineering, computing and other clinical and basic sciences for the development and application of new and enhanced imaging techniques to address problems in biology and medicine, in health and disease. Faculty and trainees pursue research in developing new imaging methods and techniques, as well as in diverse applications. The addition of a 7 tesla magnet, one of only about eight in the United States, has enabled researchers to generate images down to the molecular level and will ensure Vanderbilt remains at the forefront of research in magnetic imaging.
Some of the core areas of current interest are the development of methods for the assessment of structure, function, and metabolism including imaging in broad areas such as cancer, brain physiology, transgenic mice, cellular and molecular as well as research into the physics of imaging and spectrometry. The Institute also provides an exemplary training environment for postdoctoral fellow, graduate and medical students and undergraduates.
VUIIS Director: John Gore
Center for Human Imaging
Director: Seth Smith
Email: seth.smith@vumc.orgCenter for Small Animal Imaging
Scientific Director: Mark Does
Manager: Jarrod True
Radiochemistry
Director: Todd Peterson
Manager: Yiu-Yin Cheung
Email: vuiis.radiochem@vumc.org
VUIIS Website: https://www.vumc.org/vuiis/welcome-vanderbilt-university-institute-imaging-science
-
Vanderbilt Ophthalmic Contract Research Organization (VO-CRO)
VO-CRO provides a service platform that combines state-of-the-art technology with unparalleled expertise in ophthalmic disease modeling. This platform facilitates evaluation of the effectiveness of putative therapeutic strategies in clinically relevant models, and extends to assessment of both structure and function of ocular tissues. Our capabilities include, but are not limited to: in vitro screening for target identification/verification and mechanistic studies of drug mechanisms-of-action; in vivo rodent modeling of ocular pathologies; macroscopic and microscopic longitudinal morphological phenotyping; and longitudinal evaluation of retinal function and visual acuity.
Website: https://vo-cro.com/
-
For a list of VU Basic Sciences Shared Resources and Core Facilities CLICK HERE.