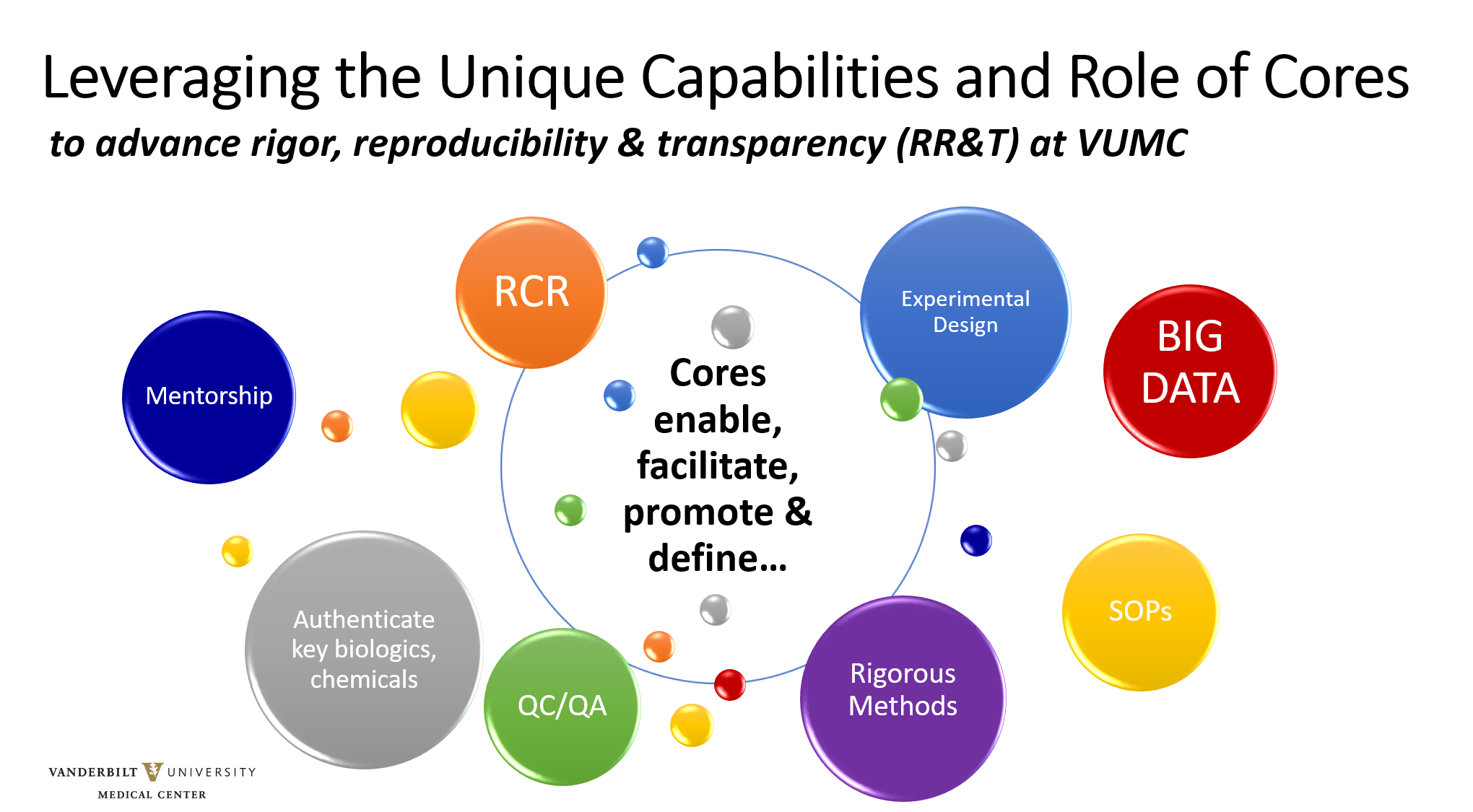
VUMC research cores and shared resources support researchers in the responsible conduct of research through training, informal mentorship and service. Cores are particularly well-suited to facilitating good experimental design and validated methods; providing authentication services for key biological and/or chemical resources; and establishing rigorous methods for acquiring and analyzing large, complex experimental data sets.
VUMC has developed guidelines for cores to promote intellectual and scholarly rigor and appropriate transparency, to encourage and enable reproducibility in science. Through the implementation and sharing of these best practices, cores demonstrate the effectiveness of a more rigorous approach to the conduct of science, enabling individual investigators to better incorporate these practices into their own research programs.
Each core has also established a data management policy to address retention and stewardship of scientific data acquired on behalf of federally-funded research programs, and is encouraged to develop other resources that support best practices of scientific rigor, reproducibility and transparency as applicable and appropriate to the core technology platforms and services.
What are Rigor, Reproducibility & Transparency (RR&T)?
- Rigor: The strict application of the scientific method to ensure robust and unbiased experimental design, methodology, analysis, interpretation, and reporting of results.
- Reproducibility: “… refers to the ability of a researcher to duplicate the results of a prior study using the same materials as were used by the original investigator…Reproducibility is a minimum necessary condition for a finding to be believable and informative.” Excerpted from: K. Bollen, J. T. Cacioppo, R. Kaplan, J. Krosnick, J. L. Olds, Social, Behavioral, and Economic Sciences Perspectives on Robust and Reliable Science (National Science Foundation, Arlington, VA, 2015).
- Transparency: Approach to sharing of knowledge and data that is as open, accessible and unbiased as possible to enable broad dissemination, understanding and utility.
What are practical ways for my group to promote RR&T? 
Review the VUMC Cores & Shared Resources Guidelines for Scientific Rigor & Transparency
This document outlines the role of cores in promoting RR&T, and lays out specific principles that should guide core operations. Review Core RR&T guidelines here.
Maintain a core-specific data management SOP
Each core should develop its own data management SOP that governs how data is defined, stored, and used within the core. The core's SOP should be easily accessible to users, so that expectations for data retention and use are clearly defined before the start of a project.
The SOP for Managing Data Retention Requirements in Core Facilities outlines best practices that cores should follow when creating and implementing a data management policy.
The SOP complements the Vanderbilt University Medical Center Policy on Sharing, Retention and Ownership of Research Data, which details institutional and investigators responsibilities related to data ownership and use. Core staff should review both documents prior to creating a core-specific data retention policy.
Host an annual advisory board meeting
OOR encourages each Core to host an annual faculty advisory board meeting. These meetings help promote transparency and provide an opportunity to reflect on ways to enhance rigor & reproducibility. OOR has prepared guidelines for these meeting, including suggested committee make-up and recommended meeting topics. Download OOR guidelines on advisory board meetings here.
Properly validate and record materials and methods
NIH recommends describing biological materials with enough information to uniquely identify the reagents, for example::
- antibodies: report source, characteristics, dilutions and how they were validated
- cell lines: report source, authentication and mycoplasma contamination status
- animals: report source, species, strain, sex, age, husbandry, inbred and strain characteristics of transgenic animals
Use of Research Resource IDs is highly recommended whenever possible - learn more below.
Partner with scientific peers to share best practices
Consider taking advantage of resources and forums provided by national and international groups such as the Association of Biomolecular Resource Facilities (ABRF). Consider publishing in journals that adhere to RR&T principles and best practices.
Provide grant-ready text that details what your core does to promote RR&T best practices
Using SciCrunch RRIDs and ABRF CoreMarketplace to support RR&T in Cores
Research Resource IDs
Similar to ORCIDs for individual researchers, Research Resource IDs (RRIDs) identify and label each component of research with a persistent, unique, citable identifier. RRIDs can be attached to Cores, as well as Antibodies, Model Organisms, Cell Lines, Plasmids, and other Tools such as software, databases, and services. In publications, RRIDs link directly to the research component cited, making it easier to identify and replicate research findings - ultimately leading to better science!
RRIDs are found in more than 1200 journals, and are required or requested in many high impact publications such as Nature, Science, and Cell. RRIDs have been linked to metrics of improved scientific quality, such as a decrease in the use of problematic cell lines.
CoreMarketplace
We encourage each VUMC core to create a listing on ABRF's CoreMarketplace. CoreMarketplace helps researchers properly cite your core in publications by assigning your Core an RRID. By facilitating data exchange between the RRID portal and CoreMarketplace, ABRF decreases the burden on Core Facilities to maintain profiles on both sites. As an added bonus, listing your facility in the Marketplace makes it easier for researchers in need of services to find your core; users can search CoreMarketplace directly, and entries are fully indexed on Google.
Once your Core has an RRID, you're encouraged to broadly advertise it on your website and in communications and presentations. Making it easy for researchers to find and cite your RRID will help your facility get appropriate credit for contributions.

The workflow above shows how RRIDs help researchers find and cite research Cores, reagents, and more. Slide from Anita Bandrowski, UC San Diego via ABRF.
How does the NIH support RR&T?
The NIH Initiative to Enhance Reproducibility through Rigor and Transparency provides resources to assist researchers in addressing rigor and transparency in NIH research and reporting efforts.
Learn more via the Rigor and Reproducibility FAQs page.
View examples of rigorous experimental design here. The NIH recommends the NC3Rs free online Experimental Design Assistant as a useful tool for designing rigorous experiments.
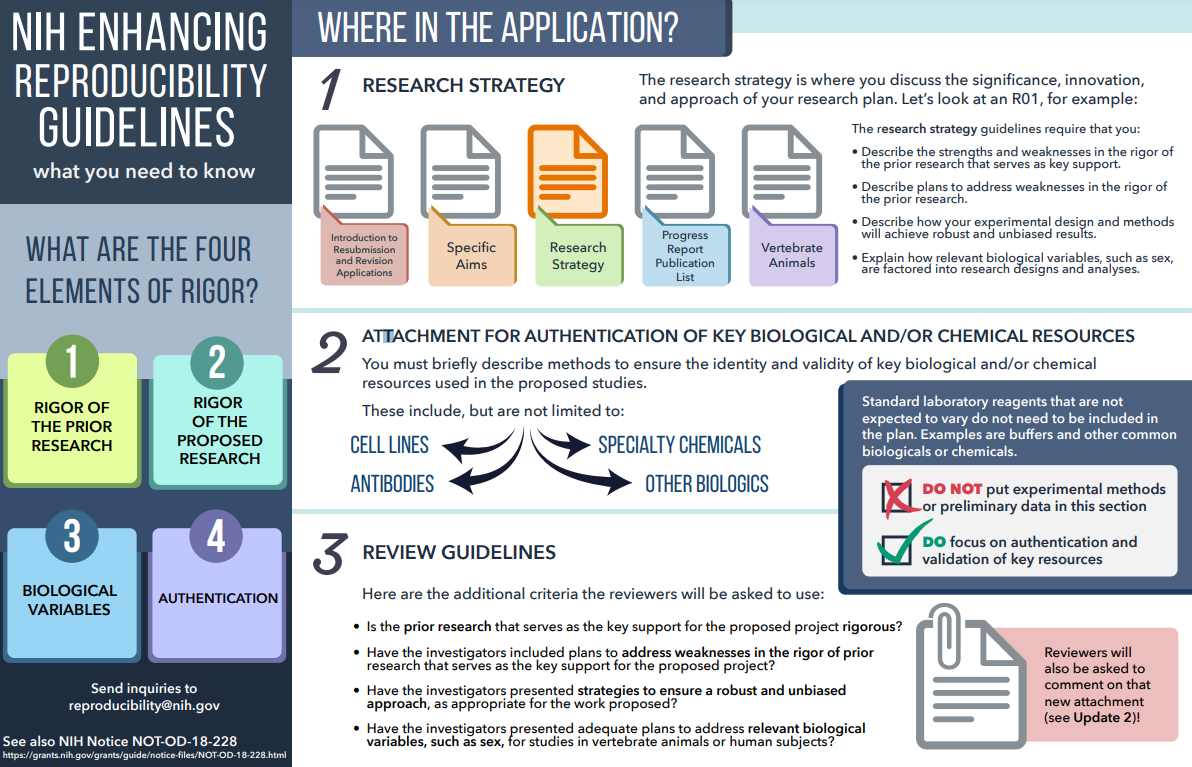
What are the requirements for authentication of Key Biological and/or Chemical Resources in NIH Grant Applications? See the Introductory Extramural Nexus blog post and Key biological and chemical resources explainer
Click image to download PDF of the NIH enhancing reproducibility guidelines.