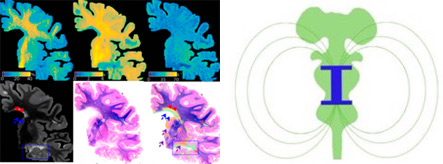
Imaging hidden mechanisms of disease in multiple sclerosis
Our work focuses on applying newly developed imaging methods to untangle otherwise hidden disease processes in multiple sclerosis. To achieve our goal, we use high and ultra-high resolution non-conventional imaging techniques both in vivo and post-mortem in humans as well as in animal models of multiple sclerosis. The mission of our lab is that of discovering pathological pathways leading to disease onset and disability progression as to pinpoint targets for new treatments. Major efforts are devoted towards the identification of imaging changes portending neurodegeneration, reflecting microglia activation, or indicating an aberrant immune system. The lab collaborates tightly with the Vanderbilt University Imaging Institute of Imaging Science, VUIIS.
Principal investigator: Francesca Bagnato, MD, PhD (francesca.r.bagnato@vumc.org)
Division: Neuroimmunology Division
Additional information: vumc.org/bagnato-lab
Translational Neurodegeneration Research Laboratory
The theme of this laboratory is to understand the biologic basis of behavior, cognition, and movement in neurodegenerative disease. Our singular focus is to develop translational therapeutics for patient populations that suffer from the devastating effects of neurodegeneration. As such, we have three main topics of research: brain glymphatic function, biologic determinants of impulsivity, and novel clinical and imaging biomarkers of disease progression. The diseases we study include Huntington disease, Multiple Systems Atrophy, Parkinson disease, and Alzheimer disease and related disorders. We also enjoy a robust clinical therapeutics research initiative, with numerous early phase clinical trials. There are many unique ways to interact with this laboratory, and interested candidates or collaborators are asked to email.
Principal investigator: Daniel Claassen, MS, MD (Daniel.claassen@vumc.org)
Division: Behavioral and Cognitive Neurology
Additional information: vumc.org/claassen-lab/
Neuroimaging and moral behavior
The Darby lab investigates the neural mechanisms underlying the most complex human behaviors at the interface of neurology, psychiatry, and philosophy, including moral behavior, delusions, and free will perception. Our lab’s overarching hypothesis is that these behaviors will not localize to a single brain region, but rather to common networks of connected brain regions that work together to bring about complex human behaviors. Our lab has worked to develop novel neuroimaging methods to test this network localization hypothesis in patients with focal brain lesions, dementia, and even psychiatric diseases. We also explore the ethical and legal implications of our research.
Principal investigator: Ryan Darby, MD (richard.r.darby@vumc.org)
Division: Behavioral and Cognitive Neurology
Additional information: vumc.org/darby-lab/home
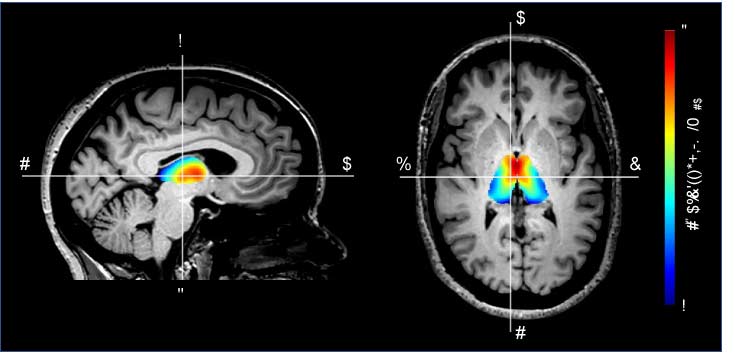
Imaging and biomarker development
Our work focuses on developing new imaging methodologies and applying these methodologies to improve our understanding of human tissue function in health and disease. A major emphasis is to identify subtle changes in physiology (e.g., biomarkers) that precede overt symptoms and clinical disease manifestations, and as such can be used to triage patients for disease-modifying therapies prior to irreversible tissue damage. Clinical applications by lab investigators and immediate collaborators focus on imaging characterization of cerebrovascular disease and stroke, HIV, post-stroke plasticity, glymphatic/lymphatic physiology, anemia, and neurodegeneration.
Principal investigator: Manus Donahue, PhD (mj.donahue@vumc.org)
Division: Behavioral and Cognitive Neurology
Additional information: vumc.org/donahue-lab/home
Computational neurogenomics
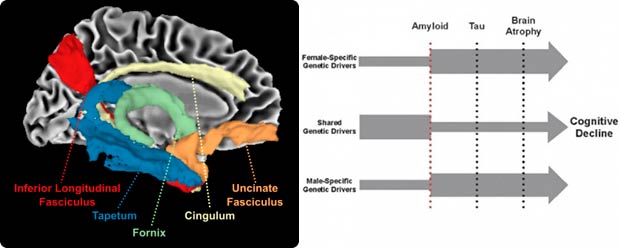
Our work is focused on Alzheimer's disease (AD) risk, resilience, and sex differences. Through transdisciplinary collaboration, we seek to facilitate a more rapid move from genomic discovery to therapeutic development. We have identified molecular drivers of resilience through genomic and proteomic analyses leveraging neuroimaging and neuropathology endophenotypes, and we integrate these diverse data types into a precision medicine approach, focusing on characterizing the best predictors of risk and resilience given an individual’s age, sex, genetic, and neuropathological context.
Investigators: Timothy J. Hohman, PhD (timothy.j.hohman@vumc.org), Logan Dumitrescu, MS, PhD (logan.c.dumitrescu@vumc.org), and Derek Archer, PhD (derek.archer@vumc.org)
Division: Behavioral and Cognitive Neurology
Additional Information: vumc.org/cnt
Disease mechanisms of Tourette syndrome
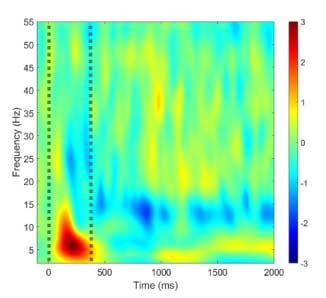
Tourette syndrome (TS) is defined by the presence of tics, but abundant evidence shows the disorder is multifaceted. Sensory and psychiatric symptoms occur in 90% of patients, and these symptoms impact quality of life to a greater extent than tics. Our research seeks to understand the interrelationship between motor, sensory, and psychiatric manifestations of TS, with the ultimate goal of identifying novel treatment targets for the disorder. Our research program involves parallel clinical and translational efforts. Clinically, we conduct cohort studies in children and adults with TS to quantify changes in tic severity, sensory perception, and mental health over time. Translationally, we use electroencephalography (EEG) to identify physiologic correlates of TS symptoms, thereby deepening insights into disease mechanisms and paving the way for clinically meaningful biomarkers.
Principal investigator: David Isaacs, MD, MPH (david.a.isaacs@vumc.org)
Division: Movement Disorders
Additional information: vumc.org/neurology/person/david-isaacs-md
Neurophysiology of consciousness and cognition
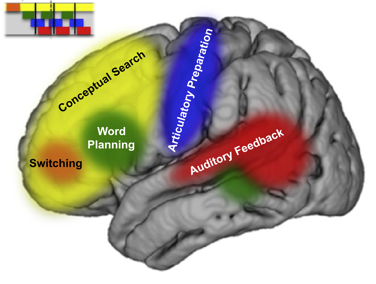
Dr. Williams Roberson’s team studies the brain activity patterns underlying consciousness and cognitive functions such as language production, perception and memory, and seeks to use these patterns to optimize the care of patients with central nervous system disorders such as delirium, dementia, epilepsy, and stroke. We use computational and quantitative analysis techniques to investigate the neurophysiological basis of cognitive functions in health and in neurologically impaired states. We are currently conducting an observational study developing quantitative EEG-based biomarkers of ICU delirium, post-ICU cognitive impairment and post-ICU psychological distress. We will use these biomarkers to identify patients most at risk for symptoms of post-intensive care syndrome (PICS), and subsequently to develop and validate targeted neuromodulatory interventions to improve cognitive and psychological outcomes in survivors of critical illness.
Principal investigator: Shawniqua Williams Roberson (shawniqua.w.roberson@vumc.org)
Division: Epilepsy & Clinical Neurophysiology
Additional information: vumc.org/neurology/person/shawniqua-williams-roberson-meng-md
Small vessel disease and cerebral amyloid angiopathy
The SchragLAB at Vanderbilt is working to better understand the mechanisms of a small blood vessel disease related to Alzheimer’s disease called cerebral amyloid angiopathy. Most patients with dementia also have damaged small blood vessels, so we hope to identify treatment targets that simultaneously improve the function of vessels and neurons. The SchragLAB also hosts the Vanderbilt Brain Bank and we are grateful to the many patients and families to have contributed to our research.
Principal investigator: Matthew Schrag, MD, PhD (matthew.schrag@vumc.org)
Division: Vascular and Cognitive Division
Additional information: SchragLAB.com
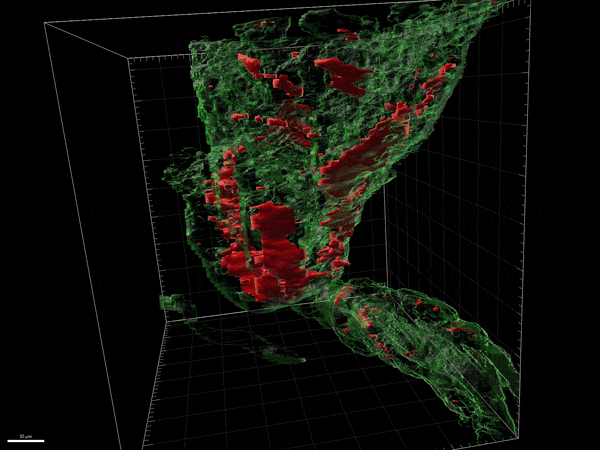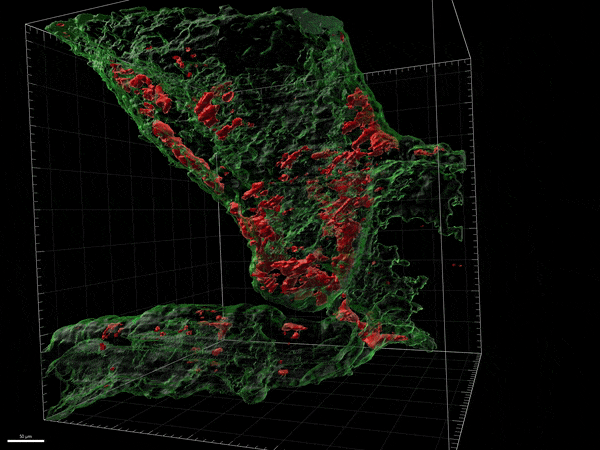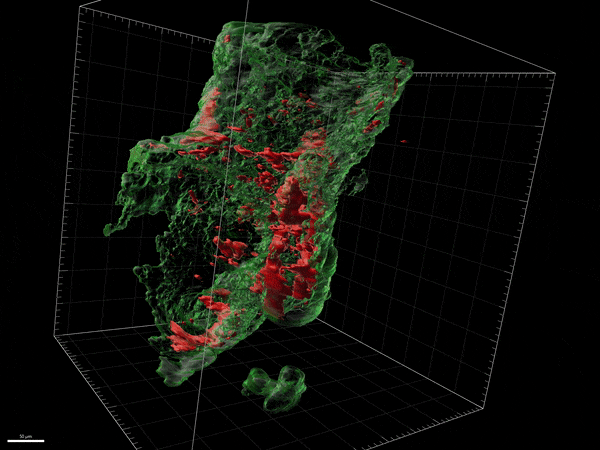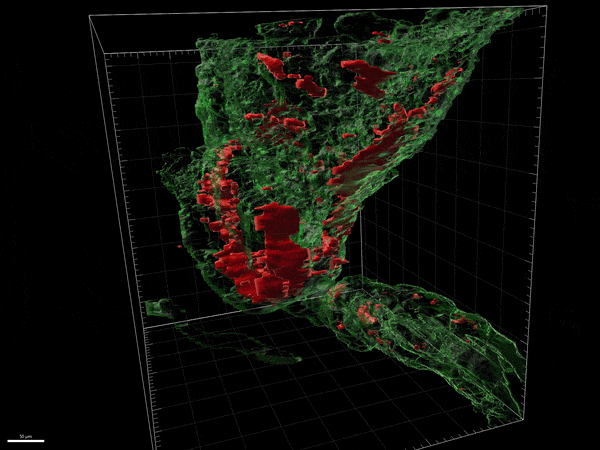
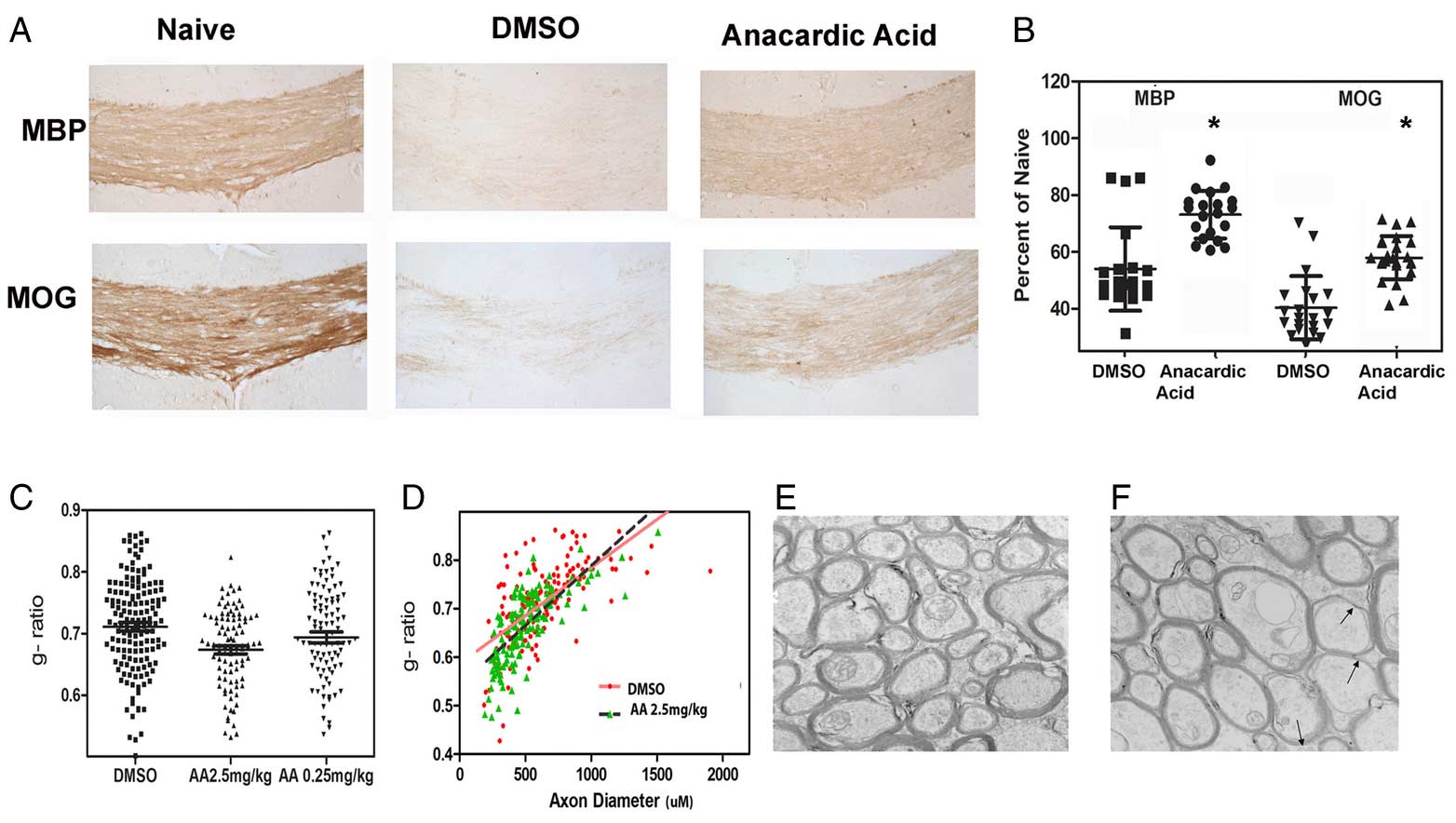
Novel remyelinating therapies for multiple sclerosis
Our laboratory is involved in the development of novel compounds that are likely candidates to promote remyelination. Destruction of myelin and the inability to recruit myelin forming cells and myelinate naked axons is one of the key features of progressive forms of MS. There are no treatments available to remyelinate naked axons and prevent their destruction. We have been examining both natural and synthetic compounds that act on myelin producing cells to increase the proliferation and maturation of myelin forming cells and eventually improve remyelination. We study the efficacy of these compounds in experimental allergic encephalitis and in Cuprizone induced model of CNS demyelination/remyelination. We have studied the mechanism of action of these compounds on myelin forming cells in culture and the dynamic changes in the brain and spinal cord in experimental model systems.
Principal investigator: Subramaniam Sriram, MD (Subramaniam.sriram@vumc.org)
Division: Neuroimmunology