CHOI LABORATORY RESEARCH
The Choi lab studies basic and pre-clinical research projects that combine multidisciplinary approaches to understand mechanisms of carcinogenic transition in gastric carcinogenesis using novel in vivo and in vitro models which are unique in the gastric cancer field.
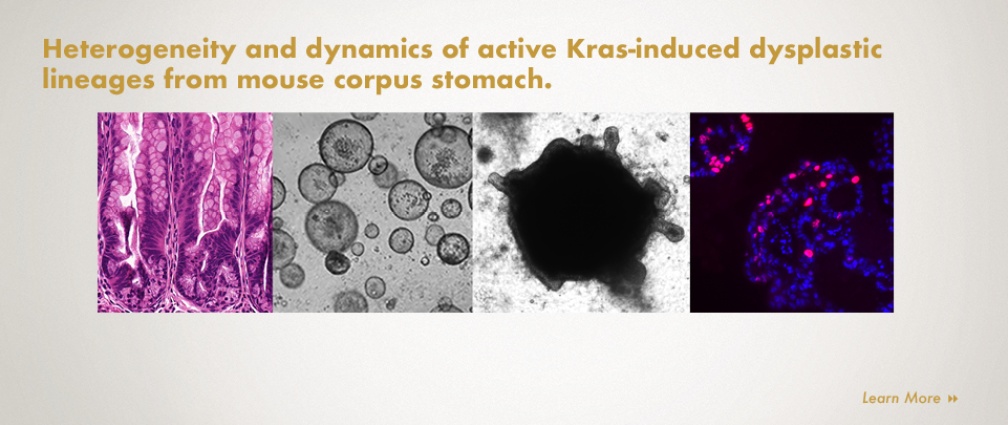
The focus of research for Dr. Choi is the comprehensive understanding of the oncogenic roles of Kras activation in gastric carcinogenesis and the functional mechanisms of carcinogenic transformation as a critical transition stage from pre-cancer to cancer. Dysplastic stage is especially considered a key transition state between pre-cancer and cancer and the dysplastic cell lineages might be critical targets for intervention in the early induction of cancer, the cellular plasticity, tumorigenic potential, or even different risks of the heterogeneous dysplastic cell lineages remain largely unknown. The Choi lab has recently established novel mouse metaplastic and dysplastic organoid lines which recapitulate the phenotypes and characteristics observed in the gastric carcinogenesis cascade, as observed in mouse models and in human gastric cancer. The organoid lines display distinguishable structures and dynamic behaviors, pathological phenotypes and molecular signatures. The Choi lab investigated cellular heterogeneity and dynamics of dysplastic cell lineages as gastric cancer initiating cells using the dysplastic organoids and also identified dysplastic stem cell populations which are possible candidates for residual stem cell populations which can drive gastric cancer. The Choi lab has also developed a new doxycycline (tetracycline)-regulatable mouse model system (rtTA) for targeted induction of responder genes only in zymogen granule-secreting chief cells which will allow us to overcome the limitations in present gastric cancer studies and have established several transgenic mouse alleles for understanding the full spectrum of gastric carcinogenesis.
The Choi lab currently investigates topics on 1) oncogenic roles of Kras activation during gastric carcinogenesis, 2) tumorigenic properties of dysplastic cell lineages and 3) cell-type specific drug efficacy and resistance in dysplastic cell lineages. These approaches will bring innovative therapeutic options for overcoming the challenges of current limitations in gastric cancer therapies, through testing biomarkers or drug treatments.

Eunyoung Choi, Ph.D.
Associate Professor of Surgery