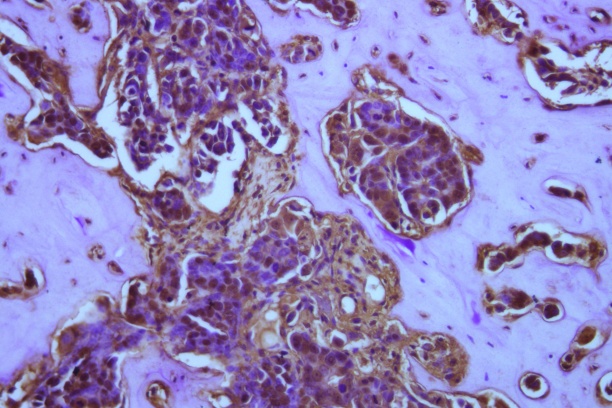
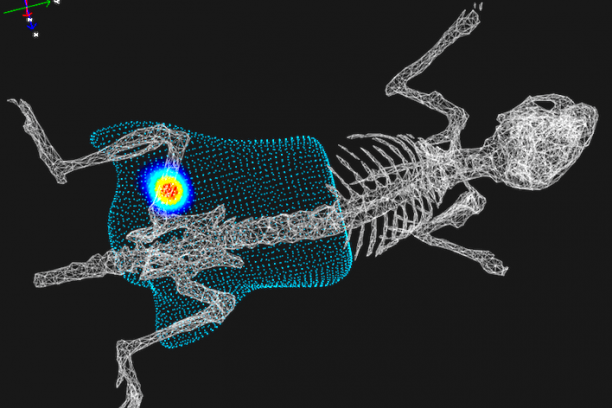
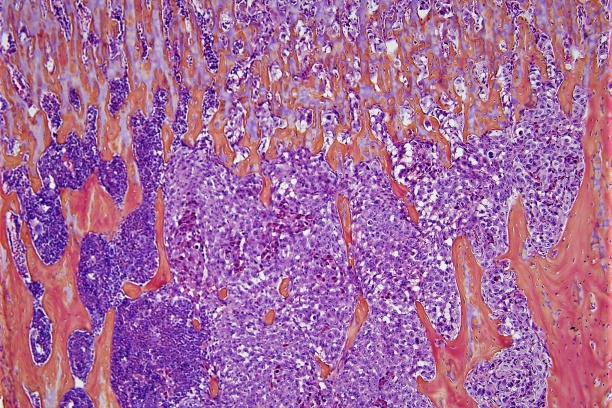
Investigating the development of the musculoskeletal system as well as diseases and conditions that negatively affect i) musculoskeletal tissue and ii) its ability to heal and regenerate, VCBB investigators aim to develop new treatment strategies and diagnostic tools that can improve the quality of life for patients with compromised bone, articular cartilage, and ligaments due to aging, trauma, infection and disease.
Jeffry S. Nyman, PhD, Director