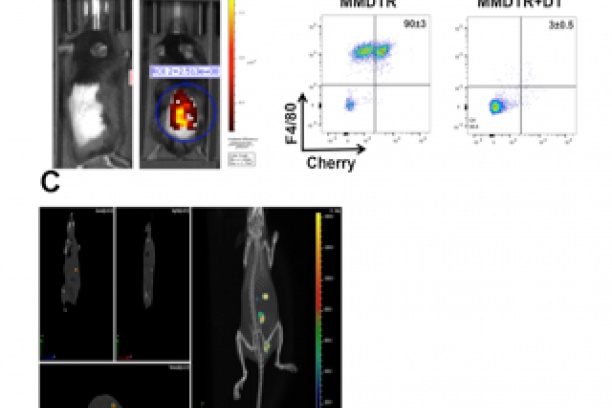
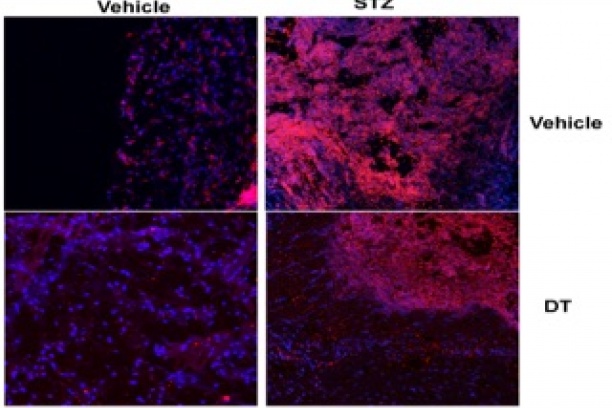
PTEN and infection
Sepsis accounts for 250,000 deaths annually in the United States alone. Unrestrained stimulation of phagocytes can induce Systemic Inflammatory Response Syndrome (SIRS) resulting in failure of multiple systemic organs. Prolonged SIRS leads to enhanced susceptibility to nosocomial infection. Phagocyte recognition of microbial products is mediated by Toll like receptors via activation of Toll/IL-1R (TIR) adaptor MyD88-dependent NFkB activation. There are numerous endogenous brakes involved in TLR activation, including phosphatases SHIP-1 and DUSP. The role of the phosphatase and tensin homolog PTEN in controlling macrophage activation and its role in controlling polymicrobial sepsis is unknown. This proposal is based on preliminary data showing that PTEN inhibits MyD88 expression by controlling actions of specific microRNAs. Furthermore, while PTEN activation has protective effects in acute sepsis, enhanced and chronic PTEN expression produces deleterious effects favoring endotoxin tolerance and possibly secondary infection.
The hypothesis to be tested is that although PTEN protects against overwhelming inflammatory responses early in acute sepsis, excessive PTEN expression is responsible for both the initiation and maintenance of sepsis-induced immunoparalysis states by impairing TLR activation in phagocytes. These studies will increase our overall understanding of how innate immunity works, and will produce insights regarding the pathogenesis of acute sepsis and enhanced susceptibility to secondary infection in deleterious conditions.
We propose the following specific aims:
- Determine how PTEN controls transcriptional and post-transcriptional modifications involved in MyD88 expression
- Determine how PTEN influences sepsis outcome and morbidities associated with secondary lung infection. The identification of specific components and their modes of action in maintenance of sepsis may identify targets for therapeutic intervention resulting in improved immune responsiveness in settings of host vulnerability, and may suggest strategies to dampen the immune response in settings of exaggerated inflammation.
TYPE 1 DIABETES AND MRSA SKIN INFECTION
Type 1 diabetes (T1D) is an autoimmune disease characterized by deficient production of insulin and hyperglycemia. Enhanced susceptibility to infections caused by methicillin resistant or susceptible Staphylococcus aureus are more common and severe in diabetics than in health people and represent a leading cause of hospitalization. It is known that phagocytes from diabetic mice exhibit poor microbicidal capacity that favors susceptibility to infection. However, diabetes is characterized by low-grade inflammation that is thought to be responsible for different co-morbidities.We have shown that insulin deficiency increases the abundance of the bioactive lipid leukotriene B4 (LTB4), which favors systemic inflammation and enhances susceptibility to polymicrobial sepsis. However, it remains to be determined the cellular and molecular mechanisms involved in enhanced LTB4 production and its consequences in skin S. aureus infection in T1D. This proposal is based on preliminary data showing that in vivo insulin receptor antagonist treatment increases LTB4 production and insulin decreases LTB4 levels and the expression of the Toll/IL-1R (TIR) adaptor MyD88; that correlates with inhibition of the microRNA-generating enzyme Dicer. Furthermore, our data show that MRSA skin infection in diabetic mice is characterized by uncontrolled lesion; with an overwhelming recruitment of neutrophils and monocytes to the skin. Treatment of diabetic mice with an ointment containing LTB4 receptor antagonist greatly improves inflammation and bacterial clearance in T1D. The hypothesis to be tested is that that low insulin drives chronic production of LTB4, which leads to excessive systemic IL1β production and actions, phagocyte recruitment, cell death, tissue damage, and ineffectual response to infection in T1D.
We propose the following specific aims:
- Determine the cellular and molecular pathways involved in enhanced LTB4 production that influence IL1β production and responsiveness in T1D.
- Investigate the role of aberrant LTB4 production on the innate response to MRSA skin infection in T1D. The identification of specific components and their modes of action in uncontrolled T1D promises to reveal novel targets for therapeutic intervention resulting in improved local and systemic immune responsiveness in settings of host vulnerability, and may suggest strategies to restore homeostasis in settings of exaggerated inflammation.