In Mozambique, in 2015, viral load (VL) testing became routine for pregnant and lactating women (PLW) who had been receiving combination antiretroviral therapy (ART) for 3 months or longer. It is very important to monitor VL in patients receiving ART in order to determine whether or not the patient is adhering to their prescribed ART regime and whether or not their treatment is effectively suppressing HIV in the body. Recent programmatic data showed an overall VL coverage rate of approximately 40% in COP18. By the end of COP19 Q1, VL coverage at FGH supported sites had risen to 49%.

To improve the requisition and clinical file availability of VL test results for patients who are eligible for routine VL monitoring, a specific intervention was designed by FGH in collaboration with the Provincial Health Directorate (DPS). The intervention was based on the introduction of a specific tool for tracking VL results from the time of the initial VL test requisition up until the results were made available in the patient’s clinical file and shared with the patient or the patient’s caregiver. The tool included the weekly monitoring of process measures such as the number of VL samples collected, the number of VL results updated in the electronic patient tracking system (EPTS), and the number of printed VL results inserted into the patient clinical files. The standard operating procedure (SOP) explaining how to properly utilize the tool and perform the necessary monitoring were shared with the district-based clinical implementation teams. In May 2019, implementation of the VL results SOP was initiated in 20 high-volume HFs in Zambézia Province. The provincial and national clinical management teams provided regular feedback on performance of weekly targets in real-time.
Namacurra Sede HF was one of the 20 HFs implementing this intervention. Results were evaluated through a cascade analysis including all steps leading up to VL results being communicated to eligible HIV-positive, ART-treated pediatric and adult patients (including pregnant and lactating women), before and after implementation of the SOP. Significant improvements were observed in each of these target groups, with the most significant improvement being noted in pregnant and lactating women, as shown in Figure 1 below:
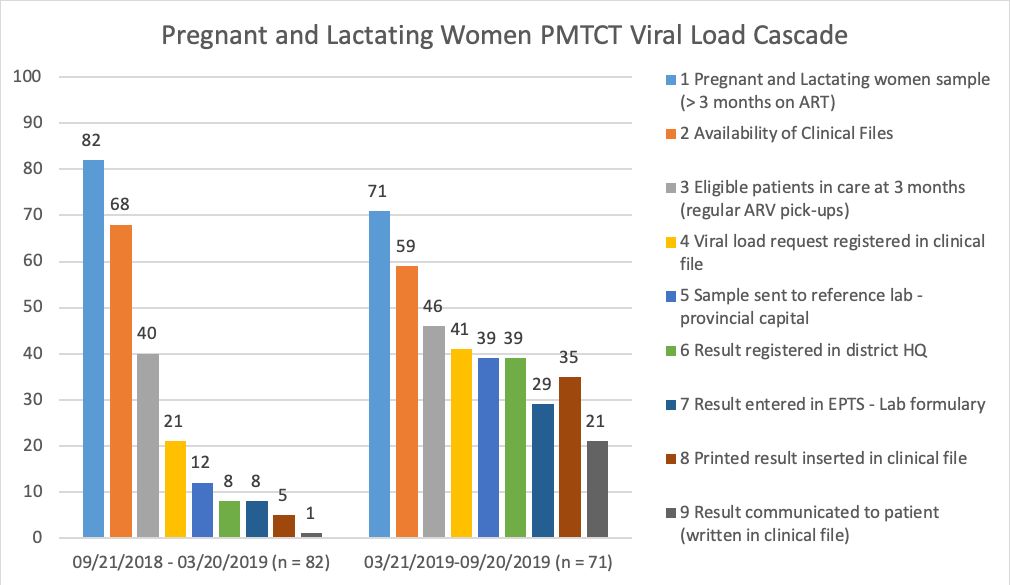
The results of the implementation of the VL results SOP led to substantial increases in i) the proportion of VL requests registered, from 52% to 89% among eligible patients, ii) the placement of VL results into available patient clinical files within the HF, from 62% to 90%, and iii) the communication of VL results to the patient or patient’s caregiver, from 20% to 60%.

As explained by Jair Fatha, the Friends in Global Health (FGH) Clinical Advisor for Namacurra District, the efficient use of the VL tracking tool helped to overcome the serious bottleneck around the insertion of printed VL results into the patient clinical files: “The laboratory would send viral loads to reception area within the HF, but receptionists did not always insert them all into patient clinical files. When we went to reception, we found a lot of results were not included in the clinical files. As a result, the patients came to the appointment and left without knowing their results, and the clinical staff did not follow-up to see if the viral load results had arrived or not from the laboratory. This tracking sheet really helped us to ensure that the patient's result was entered into their file.”
According to Daniel Mundiva, the Quality Improvement (QI) Focal Point for the Namacurra Sede HF, “Before the tracking sheet was introduced, fewer people knew their viral load result. There was a long waiting period to learn the result (…). Now with the tracking sheet, if a patient does not have the result, we can track where the patient’s viral load result is. If reception of the result is delayed, we can easily identify the reason.” Daniel emphasized that “Thanks to the tracking sheet, we have almost 100% of VL results returned, hardly any misses. This facilitates the monitoring of patients”.
António Fiscal, the Director of the Namacurra Sede HF, ends by stating: “The introduction of this tracking sheet is of added value.”